[Part of a series: The Age of Steam]
A New Synthesis
In the eighteenth century, new lines of communication and new alliances were forming between the world of the artisan and craftsman on the one hand, and the world of the “schoolmen,” the university scholars, steeped in abstract knowledge, on the other. This convergence arguably began with the engineers of fifteenth-century Italy, the Renaissance men par excellence such as da Vinci and Brunelleschi, but reached a far higher pitch several centuries later as scientific learning seeped into popular culture.
We have already seen that the natural philosophy of Galileo and his ilk had a more practical bent then that of their predecessors. By the eighteenth century, this new breed of philosophers had established their own institutions in Britain and France – the Royal Academy and the Royal Society. They had also acquired a great deal of cultural prestige, especially after the dramatic unification of terrestrial physics and astronomy carried out by the most exalted of their company, Isaac Newton. No Aristotelian philosopher had ever prompted laudatory couplets from a poet with the stature of Alexander Pope:
Nature and nature's laws lay hid in night; God said "Let Newton be" and all was light.[1]
With their increased status, the new philosophers harbored serious hopes of carrying out the ambitions of Sir Francis Bacon, to command nature by obeying her. Bacon recognized three great transformative inventions introduced to Europe since antiquity – gunpowder, the compass, and the printing press. But these achievements, he argued, had been the mere result of time and chance. How much more consistent and potent control over nature might man develop after acquiring a deep and thorough understanding of its natural laws?
To advance the practical arts through science necessarily required a serious study of their present condition, and this the French Academicians provided in their Descriptions of the Arts and Crafts, appearing in 113 volumes starting from 1761. The original editor, René-Antoine Ferchault de Réaumur, placed the entire work clearly within the tradition of Bacon’s project in his introduction:
What new degrees of perfection in the arts may not be expected if the scholars who have acquired knowledge and experience in the various departments of natural science give themselves the trouble to examine and to explain the often ingenious labors undertaken by the skilled man in his workshop? …The surveyor, the engineer, the chemist will give new insight to an intelligent craftsman so that he may overcome obstacles which he himself has not ventured to remove.[2]

This dream, that the noodlings of robed academics could invigorate the productive engine of human society, invited skepticism, or even mockery. Jonathan Swift satirized the savants of the Royal Society in Gulliver’s Travels through his Academy of Lagado, whose inhabitants carried out ridiculous projects such as extracting food from human excrement and making clothing from spider silk. But despite such criticism, by the middle of the eighteenth century the increasing interaction between practical and scientific thought had begun to pay real dividends, in the form of a new kind of scientific, analytical approach to the crafts.
One sign of this was the emergence of the profession of civil engineering. In Britain, its foremost practitioner was John Smeaton, born in 1724, a few years before Newton’s death. Though his father had intended young John to follow him into the law, he instead set out on a career in London as a maker of philosophical instruments: scientific, mathematical, and navigational apparatus, such as telescopes, astrolabes, sextants, and air pumps. The growing European trade in this kind of equipment was another mark of the increasing degree of contact between scholarly knowledge and craft knowledge. Buyers included not only practicing surveyors, navigators, and natural philosophers, but also gentleman dilettantes. Popular treatises such as The Microscope Made Easy (published in 1742), further stimulated demand for scientific curiosities among middle-class buyers.[3] Touring lecturers — whom Smeaton once referred to as “the common herd of conjuring Philosophers about Town” — also performed demonstrations for a scientifically-curious public, and were always in need of instruments. Smeaton himself hobnobbed with a less common herd of philosophers, as a member of the Royal Society.
But Smeaton soon took an interest in larger constructions, such as waterwheels, bridges, lighthouses, and canals. He called himself a “civil engineer” (in contrast with the engineers who constructed roads, bridges and fortifications for military purposes), and founded the Society of Civil Engineers in 1771, at a tavern in London. Smeaton threw himself into this newly invented profession with indefatigable energy. His engineering reports, some commissioned and others pursued out of his own interest, fill several published volumes. He took a hand in virtually every major public works project in Britain in the second half of the century. Most often the local authorities called upon him to study the improvement of their region’s waterways – the drainage of wetlands, the cutting of canals, or the building or repair of locks, dams, or bridges. His clients, however, often failed to follow through on his advice, perspicacious though it may have been, for lack of funds to carry it out.[4]
Smeaton analyzed the construction of machinery in Newtonian terms that would be familiar to a high school physics student of today:
…if the weight raised is multiplied by the height to which it can be raised in a given time, the product is the measure of the power raising it… if a power can raise half the weight to double the height; or double the weight to half the height, in the same time another can, those two powers are equal.[5]
Smeaton wrote these words in a study on the improvement of water and wind mills, a line of research that earned him the Royal Society’s Copley Medal. That this award from a philosophical society had been granted for the investigation of practical machinery was a sign of the times. Smeaton’s approach to craftsmanship fused handicraft tradition, thorough quantitative analysis, and the theoretical frameworks of the new philosophy.[6]
Unsurprisingly, the ubiquitous Smeaton eventually turned his attention to the inefficient Newcomen engine. Following his modus operandi for improving the waterwheel, in 1765 he designed a small model engine (with an eighteen-inch diameter cylinder) on which to experiment. He calculated the load on the piston and the power delivered by the engine, and devised ways to improve the seals and the working of the valves. The culminating achievement of this work was an engine he designed for the Wheal Busy mine in Chacewater, Cornwall, that replaced two prior engines. Erected in 1775, it generated 72 horsepower, against the combined 60 of its predecessors, while consuming roughly 60% as much coal.[7]
Smeaton’s careful quantitative studies enabled the steam engine to be used more widely and efficiently. But they did not change the fact that the steam engine had few applications beyond pumping water out of mines, and remained expensive even at that, except at the pit head of a coal mine. To make a qualitative transformation in the status of the steam engine would take the more imaginative work of another maker of philosophical instruments, with contributions from a branch of physics little touched by Newton – the physics of heat.
Latent Heat
Some historians have expressed the sentiment that “the development of the steam engine took place with little support from science and… that the steam engine did more for science than science did for the steam engine.”[8] It is certainly true that, nearly two centuries on from Bacon’s masterwork, the Novum Organum, his program had failed to generate practical results at the scale that he had hoped for. Despite the increasing influence and status of natural philosophers across eighteenth-century Europe, they were not producing new inventions by the dozen with the same import as gunpowder and the printing press. Nonetheless, it is difficult to sustain the argument that the technical development of the steam engine owed nothing to abstract knowledge of nature. If the study of pressure made the pumping engine of Newcomen possible, the study of heat allowed it to become far more efficient and thus opened up a far wider range of potential applications.
Traditional, Aristotelian natural philosophy treated heat as a quality of a substance, alongside such things as its color or wetness. Qualities could differ in degree or intensity, but, unlike such measurable characteristics as weight and extension, they could not be quantified.[9] This understanding of heat had to be thrown out the window after the introduction, in the late sixteenth and seventeenth centuries, of devices for measuring temperature – first thermoscopes (which lacked a scale), then true thermometers. However, if the science of heat could no longer rest on Aristotelian qualities, great confusion remained about just what it did consist of.
Galileo posited that heat consisted of motion – the faster the motion, the greater the heat (and therefore temperature). However, he associated this motion not with normal matter, but with special fire particles within matter. His was thus an admixture of what later came to be seen as two distinct theories of heat – a motive theory, which attributed heat to the motion of matter at a tiny scale; and a fluidic one, which attributed heat to a subtle or imponderable fluid that flowed within matter.
Bacon had championed the motive theory, and used it as his canonical example for the derivation of new knowledge from the experimental investigation of nature. After examining numerous facts about heat, he concluded:
From the instances taken collectively, as well as singly, the nature whose limit is heat appears to be motion. This is chiefly exhibited in flame, which is in constant motion, and in warm or boiling liquids, which are likewise in constant motion. It is also shown in the excitement or increase of heat by motion, as by bellows and draughts… It is further shown by this circumstance, namely, that every substance is destroyed, or at least materially changed, by strong and powerful fire and heat: whence it is clear that tumult and confusion are occasioned by heat, together with a violent motion in the internal parts of bodies; and this gradually tends to their dissolution. …it must not be thought that heat generates motion, or motion heat (though in some respects this be true), but that the very essence of heat, or the substantial self of heat, is motion and nothing else…[10]
But despite this endorsement of the patron saint of British experimental philosophy, the opposing fluidic theory was far more popular in the eighteenth century. It could explain clearly why heat flowed from warm to cold bodies – just as a fluid would flow from a full to an empty container – and why heat was conserved – since the amount of fluid in the system did not change. Moreover, it fit neatly with other theories of the day – natural philosophers also understood electricity, for example, as a subtle fluid.[11]
Contention remained, however, on the question of determining how much heat an object could contain. If heat were a fluid, heat and temperature could not be quite the same thing; the iron within a massive steam engine surely contained more heat than a single iron nail raised to the to the same temperature. Hermann Boerhaave stated in his Elements of Chemistry (1732) that the quantity of heat was directly proportional to the volume of heated matter, and that the thermometer measured the density of the fluid; other philosophers argued that heat was proportional to mass. Unfortunately, the data confounded both of these positions. Daniel Fahrenheit performed an experiment in which he found that mercury and water reached an equilibrium exactly between their two initial temperatures when they were mixed at a ratio of three volumes of water to two of mercury. The volume theory predicted that this should have required only two volumes of water, the mass theory thirteen volumes (since mercury is far more dense).[12]
Joseph Black, Professor of Anatomy and Chemistry at Glasgow College in Scotland, was the first to untangle this problem. It was not unusual in the eighteenth century for someone like Black, with medical training, to work on problems in chemistry and the physics of heat. (Boerhaave, too, was a physician.) All of these fields were seen as closely allied – the dysregulation of the body’s chemistry and heat were believed to be major sources of illness, chemistry was essential to the making of pharmaceuticals, and the application of heat was required to induce many kinds of chemical transformation. Chemical science also had many applications in industry; many trades relied on alkalis like potash. Indeed, along with instrument making and civil engineering, chemical industries such as dye-making were at the forefront of the fusion of philosophical and practical knowledge. Black was frequently called upon to consult on industrial processes as varied as the refining of sugar, the glazing of ceramics, and the making of vinegar.[13]

His investigations into the fluid temperature problem began with the observation that water does not liquify instantly upon reaching the melting point: if it did so, massive flash floods would scour away the settlements along every waterway below any snow-bearing mountains every spring. Only after absorbing a significant amount of heat from the air do the snows (or ice) begin to melt. But during this time, the temperature of the snow does not change at all. So where does the heat go? Black called this missing heat, stored somehow within matter without changing its temperature, “latent” heat, and he was able to measure it. The amount of heat required to melt a given mass of ice or snow would raise the temperature of the same amount of liquid water by 140 degrees (Celsius). He found that the same applied to the transformation of water into steam, and his experiments showed that turning a unit of water into steam required a staggering 810 degree-equivalents of heat.[14]
His investigations of latent heat led Black on to the idea of heat capacity: that each type of material has a different affinity for heat which allows it to absorb more or less of it. A given mass of water absorbed a great deal of heat to change in temperature by degree, mercury far less, explaining the result of Fahrenheit’s experiment.[15]
This re-conception of heat as a something quite distinct from temperature provided the backdrop for the reinvention of the steam engine. Black’s experiments to measure the latent heat of steam took place in 1764, just as a young instrument maker named James Watt, employed by the very same college where Black worked, was beginning his own investigations into the workings of Newcomen’s engine.
Watt
James Watt was born in 1736 in the small town of Greenock, Scotland. It lay about twenty-five miles down the Firth of Clyde from Glasgow, for which it served as a custom-house port. The second city of Scotland had begun a growth spurt in the eighteenth century, fueled by trade with Britain’s overseas empire in cotton, sugar, and especially tobacco – by the 1770s, Glasgow imported some 47 million pounds of it a year for the smokers and snuffers of Europe. The population doubled in the first generation of Watt’s adulthood, from 23,000 in 1755 to 42,000 in 1780, and would nearly double again by 1801, though by that time the American Revolution had smothered the tobacco trade.[16]
The young Watt worked for a time for his father – a well-to-do carpenter, shipwright, and merchant – before being attracted, liked Smeaton before him, away from the family business and into the instrument trade. In a way, however, he was carrying on an ancestral tradition: the previous two generations of Watt’s family had also blended interests in mathematical abstraction and practical crafts. His paternal grandfather, Thomas Watt, who moved to the Greenock area in seventeenth century, taught mathematics and navigation. Thomas had two sons – one, James’ uncle, worked as a surveyor but died young; the other, James’ father, dabbled in the repair and maintenance of nautical instruments as part of his ship-building business.[17]
In 1755, at age 19, Watt set out for London, where he secured a year’s apprenticeship under John Morgan for a fee of twenty guineas. The bespoke nature of Morgan’s business ensured that Watt remained constantly busy and constantly learning. He made rules for measuring distances, sectors for calculating proportions, theodolites for surveying territory, and quadrants and compasses for navigation. When he returned to Scotland in 1756, Glasgow College hired him to clean and restore a set of instruments recently donated by a Jamaican merchant. He executed the task with skill enough to be subsequently hired as the official instrument maker of the college.[18]
Watt established a good working relationship with Glasgow College’s professors, several of whom often frequented his workshop, including John “Jack” Anderson (the College’s professor of natural philosophy, who had encouraged Watt to go to London for his training), Adam Smith (who would soon publish his Theory of Moral Sentiments), and Joseph Black (with whom we are already familiar). Black ordered a number of items from Watt, including a steam digester, invented by Denis Papin, another steam pioneer, a century earlier. With Anderson’s assistance, Watt was also allowed set up his own commercial storefront on the ground floor of the college premises, to help him earn his keep in his trade.
The shop became a popular hangout for professors and students, and Watt gained a reputation with his visitors as a thoughtful philosopher as well as a skilled mechanic. But he did not have a knack for business – he recoiled from self-promotion and the management of workmen – and he still relied on allowances from his father to get by.[19] Business seemed to pick up by the early 1760s after he formed a partnership with John Craig, a well-capitalized and business-savvy Glasgow merchant. They set up a new, larger shop on the Trongate, a fashionable shopping district. However, the success was illusory – Watt required steady infusions of cash from Craig to keep his business afloat.[20]

In all probability, Watt’s story should have ended there. A bustling instrument maker of modest and precarious prosperity, the documentary traces of his life might have provided fodder for some future doctoral student’s dissertation, but it was never likely that he should have become the namesake for a unit of measure. But then came the steam engine. Watt first turned his attention to it, briefly, in 1759. A student at the college, John Robison, who frequented Watt’s shop, came to his friend with an idea for a steam-driven carriage. Watt attempted to build a model of such a conveyance, without success, and Robison shipped out on a naval vessel as tutor to an admiral’s son shortly thereafter. But something about the steam engine tickled Watt’s mind, and he continued to ponder on it. He studied literature on the steam engine, especially the writings of John Desgauliers, a fellow of the Royal Society and successful public lecturer on natural philosophy, who had made his own improvements to Savery’s engine.
A new opportunity arrived in 1763, when Professor Anderson asked Watt to repair the model steam engine owned by the college, an adorable little thing with a cylinder just two inches in diameter and a boiler the size of tea kettle. Anderson had requisitioned it from a London instrument maker, Jeremiah Sisson, but it had never worked as it was supposed to. After working up a head of steam, it would run for a few strokes, then stop.[21] This broken toy steam engine, of such little apparent significance, got under Watt’s skin; it was a thorn in his paw, a burr in his sock; he could not leave off picking at it. He would not quit until he had solved the puzzle of why the model failed to work as it should, and solving that puzzle would lead to him to reinvent Newcomen’s engine.
The Engine Unleashed
In scaling down a Newcomen engine, Sisson had made the cylinder proportionally far thicker relative to its diameter than on a true engine.[22] This made it the perfect vehicle for illuminating the limitations of Newcomen’s design. Watt quickly discovered that a single stroke of the model consumed several times the cylinder’s volume in steam, and therefore the boiler literally ran out of steam, causing the engine to grind to a halt. The relatively large amount of metal in the cylinder was exhausting the engine, because it required a great deal of heat to bring it back to a temperature that could support steam after being cooled by the condensing cycle.

Watt also noticed another strange thing. It required injecting a surprisingly large quantity of water to condense the steam for the power stroke. Mixing boiling water with same amount of cool water produced tepid water, as one would expect from a traditional mixing rule like Boerhaave’s, but steam at the same 212-degree Fahrenheit temperature seemed to have extraordinary powers to heat water. To explore this phenomenon, Watt devised his own experiment. He set up a glass cylinder of well water and a steaming kettle, and connected them with a glass tube. He then watched until the level of the well water ceased to rise (having itself reached the boiling point, it was unable to condense any more steam). He found that it had increased in volume only by 1/6th – in condensing to a liquid, the steam was able to increase 6 times its own weight in water to the boiling point. Watt turned to his friend Black for an explanation, and learned of Black’s theory of latent heat.[23]
Watt realized that he now had the understanding needed not just to fix the model engine, but to overcome the fundamental inefficiency of Newcomen’s engine. But he found himself on the horns of a dilemma. To reduce fuel consumption required reducing the amount of heating and cooling of the metal cylinder. And Watt could now compute the exact amount of water required to condense the steam in the engine without cooling the walls around it much. But making this adjustment enfeebled the engine, it could only generate a tiny amount of power. Again, Watt turned to his knowledge of natural philosophy to understand why – in 1757, William Cullen, Black’s mentor, had written up a report on his experiments with boiling liquids, showing that water boiled at a lower temperature in vacuo. He surmised, therefore, that his still-hot working cylinder was re-boiling some of the the condensed water back into steam, preventing a strong vacuum from forming inside the engine, and therefore counteracting the force with which the atmosphere depressed the piston.
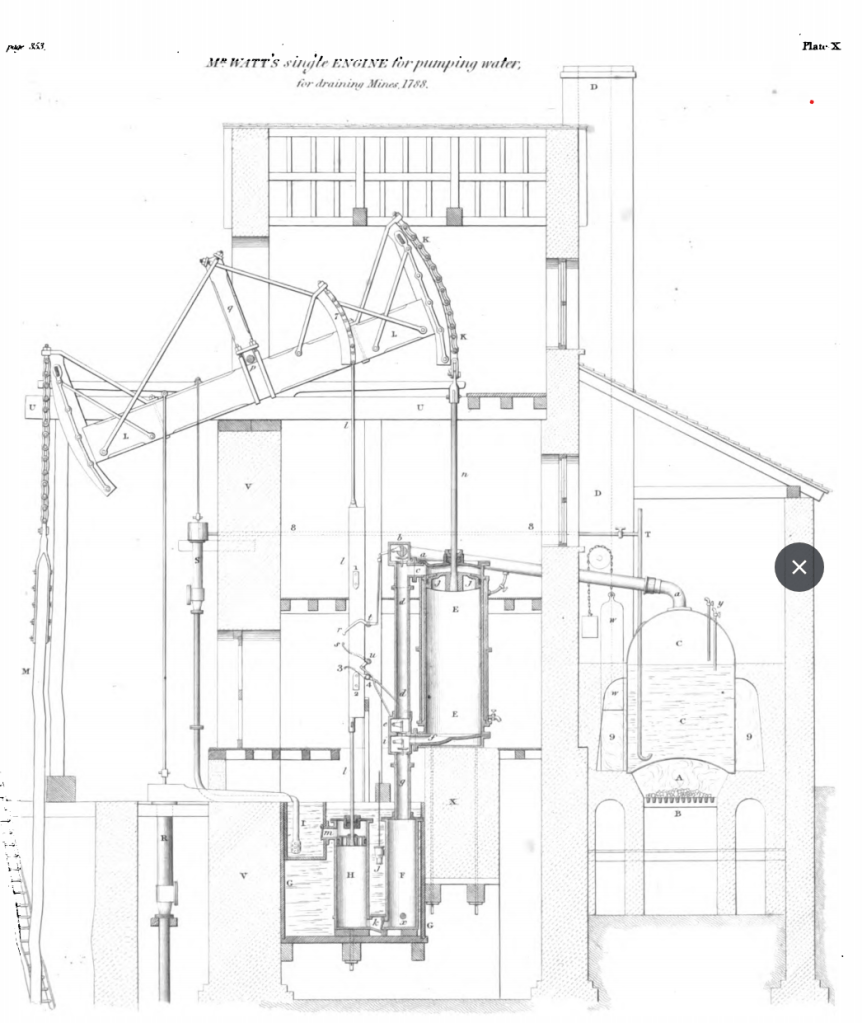
So, what to do? Keeping the cylinder hot made the engine too weak to be useful, and cooling it down wasted a huge amount of fuel to re-heat it so that it would hold steam again on the next stroke. The answer struck Watt one day in 1765 while he was out for a Sunday stroll on the Glasgow Green – separate the part of the engine responsible for generating power (which needed to stay hot), from the part responsible for condensing the steam (which could stay cool). The new chamber, called the “condenser”, would be connected to the cylinder by a pipe, with a valve between them. Once the main cylinder had filled with steam, the valve would open, allowing steam to flow into the evacuated condenser. There it would come in contact with cool water and turn to liquid, creating a vacuum in the combined condenser/cylinder system, which allowed the piston to drop. Finally, the engine’s machinery would close the valve again and the process would repeat. You may recall that Newcomen had improved on Papin’s steam engine design by separating the boiler from the piston; in a sense Watt’s innovation was a further extension of this process of disaggregating the logical components of the steam engine cycle into physical components.
The separate condenser opened up another new possibility – because the cylinder was always kept hot, it was now possible to introduce steam above the piston at the same time that the steam beneath was being condensed, providing additional downward pressure above and beyond the natural air pressure, and so increasing the power and efficiency of the engine. This would also keep cool air out of the interior of the engine, helping the cylinder stay hot. By encasing the whole cylinder in a steam-filled jacket, Watt would keep cool air off of its exterior as well. At the same time, however, the new design also created new technical challenges. The condenser had to be emptied of water at each cycle and also pumped free of any air that leaked in – otherwise it would not contain the vacuum necessary to draw the steam in. Another vexing problem was sealing the piston. Newcomen had sealed the vacuum below the piston off from the air above it with a leather seal atop the piston, which in turn held a thin layer of water atop it. However, a perpetually hot cylinder with steam intentionally introduced into its top end could no longer sustain a puddle of liquid water.
Boulton & Watt
What Watt needed most now was money, money to build a full-sized model and work through the obstacles that lay between the idea of a separate condenser and the realized fact of a working and salable steam engine. But his partner and financier, John Craig, had recently died. So, Watt turned instead to Joseph Black, who loaned Watt some money, but, more importantly, introduced Watt to the industrialist John Roebuck. Black knew Roebuck from his chemical consulting work, as Roebuck made sulfuric acid for use in dye-making and other industrial processes.[24] Roebuck’s largest business concern, however, was his ironworks at Carron near Falkirk, Scotland. It made, among other things, steam engines and cannons (both of which required boring out an iron cylinder), and gained fame in the British Navy for its short “smasher” cannon, which became known as carronades. Roebuck also leased a coalfield at Borrowstounness (or Bo’ness) a few miles to the east of his ironworks, with the expectation of acquiring a cheap supply of fuel. But he found that the mines were constantly flooded, and a Newcomen engine lacked the power to overcome the inflow of water. Thus Roebuck had a great interest in the possibility of a more powerful and efficient steam engine. In 1768, he agreed to take over Watt’s debts, help him file a patent, and provide him with a workshop at a house in Kinneil, in exchange for two-thirds ownership of his work.[25]
On his way back from filing the patent paperwork in London, Watt stopped over in Birmingham, and visited the well-known Soho Manufactory of Matthew Boulton, which made small metal “toys”: buttons, watch chains, belt buckles, and so forth. Boulton, about eight years older than Watt, was a well-established businessman, one of the most successful in Birmingham. But he was in great need of a new power source to drive the machinery at his works. His water supply often failed in the summer, forcing him to resort to an expensive horse-driven mill. Like Watt, Boulton had imbibed the popular scientific culture of the day. He had read up on physics and chemistry, and performed experiments with electricity. He was therefore not reluctant to dive into the technical problems of power supply himself. He even had his own model steam engine built, and requested advice from Benjamin Franklin (then resident in London as a representative of the American colonies) on its improvement.

Nonetheless, he was impressed by Watt’s ideas and work, and decided to leave off his own experiments and see what came of the Roebuck and Watt partnership. Watt, always in need of more funds, hoped to make Boulton a third partner, a hope that became ever more desperate as Roebuck slid towards financial ruin under the weight of his failed coal mine and a European credit crisis. But Boulton demurred, partly because of his own commitments and financial problems, and partly because Roebuck refused to allow him in as a full, equal partner. Finally, in 1773, Roebuck’s affairs collapsed into bankruptcy. For Boulton, who happened to be one of Roebuck’s creditors, the time was ripe to take a chance on Watt, and waived the money owed him in exchange for Roebuck’s interest in Watt’s patent. The following year, Watt moved his workshop south from Kinneil to Birmingham.

If anything proves Edison’s claim about the ratio of inspiration to perspiration required in works of genius, it is Watt’s steam engine. Nearly a decade had passed since Watt’s Sunday stroll. He had invested thousands of pounds in the engine (albeit much of it Roebuck’s), given up instrument making and taken up a career in civil engineering to pay his bills, borrowed yet more money from Black, lost his only steady job, as engineer for the Monkland Canal, and lost his beloved wife Peggy in childbirth. Yet the engine still didn’t work to Watt’s satisfaction.[26] The most serious problem was the seal around the piston. Much like Edison in his exhaustive search for a material for his light bulb filament a century later, Watt tried cylinders of tin, wood, copper, and cast iron; pistons made of leather, cork, and asbestos; sealants of horse dung, vegetable oil, and tallow.[27]
In addition to trial and error, Watt continued to apply quantitative methods to his work. The degree to which abstract knowledge had penetrated the world of crafts is evident in the 1797 encyclopedia article “Steam Engine”, written by Watt’s old friend John Robison, which is riddled with tables and equations. It describes, for example, the use of the “hyperbolic logarithm” (what we would call a natural logarithm) to compute the total pressure on the piston during the expansion of steam, a computation first performed by Watt himself. Yet, try and calculate as he might, a gap always remained around Watt’s piston through which steam could leak.
Watt’s problem, ultimately, was one of precision, not materials. Boring machines for cannon and steam engines worked by turning a steel cutting blade (driven by a water wheel) and pressing it into a cylinder with a hole cast down the center. This method had two problems – the blade would tend to follow any irregularities in the casting, and the weight of the blade would cause it to cut more metal from the bottom of the cylinder than the top. Smeaton developed an improved boring machine for Roebuck’s Carron works that would make a more truly circular hole, but not necessarily a straight one.
The final piece fell into place for Watt in 1775, when Boulton requisitioned an iron cylinder from John Wilkinson, who had devised a new boring technique. He cut into a solid cylindrical casting with no pre-existing hole, and placed his cutting blade on a rigid iron bar, supported on both ends. A rack and pinion advanced the cutting head down the bar, ensuring a true, straight cut.[28] Wilkinson’s cylinders, true to a circle with a variation of less than width of a shilling — about 1/10th of an inch — were sufficiently precise that Watt’s pistons could work without leaking a noticeable amount of steam.
While Watt concentrated on the technical refinement of his engine, Boulton addressed himself to matters of business and politics. Boulton had broader ambitions for the use of Watt’s intellectual property than just improving his own works at Soho; he harbored visions of a global commercial empire of steam. In a 1769 letter to Watt, before they had formed a partnership, he wrote of these nascent plans:
To … produce the most profit, my idea was to settle a manufactory near to my own, by the side of our canal, where I would erect all the conveniences necessary for the completion of engines, and from which manufactory we would serve all the world with engines of all sizes…[29]
To that end, he wanted to crank out engines like buttons in the Soho Manufactory, producing them en masse through the rigid division of labor. This did not prove practical – engines were still bespoke items that had to be erected on site, and Boulton & Watt relied on several suppliers to produce major parts outside the Soho works (Wilkinson’s cylinders, for example). Nonetheless, he was able to build up a somewhat distributed local production system sophisticated enough to produce over 400 engines by 1800 – roughly one every three weeks. To further ensure his future profits, he dispatched Watt to London to argue (successfully) in favor of a special bill in Parliament that would extend his patent rights for twenty-five more years. Shortly thereafter, in June 1775, he formally created the legal partnership of Boulton & Watt.
One of the first engines from Boulton & Watt went to a traditional steam engine customer – Bloomfield Colliery, in the coal fields northwest of Birmingham. At thirty feet tall, with a 50-inch diameter cylinder, and a price of £2000, it dwarfed anything Watt had previously built.[30] Another went to John Wilkinson, to work the bellows of his iron foundry, which sat just across the River Severn from Coalbrookdale, about thirty miles northwest of Boulton & Watt’s manufactory at Soho. The greater efficiency of Watt’s engine had opened up new applications for steam, and the cycle of iron, steam, and coal through the Birmingham region – already a major metalworking center – grew even more intensive.
Customers came calling from further afield as well – the metal miners of Cornwall, who sorely needed help to keep their mines clear of water, but who had found limited success with Newcomen engines, were eager for a better solution. In 1777, Watt himself went to supervise the construction of the first of Boulton & Watt engine in Cornwall, at the Wheal Busy copper mine, the very same mine where Smeaton had recently installed his own engine. Watt, now over forty, had become a rather fussy man of middle age, and found the manners of the Cornwall mine owners vile and their skeptical attitude toward his work infuriating. Nonetheless, he and his workmen got the engine running, and it shamed all the doubters when it “forked” (in the argot of the miners) more water than a traditional engine at one-third the cost in fuel. Even while consuming less coal, Watt’s engine generated roughly three time as much power as a traditional Newcomen engine, and twice as much as Smeaton’s best design. Working day and night at a steady fourteen strokes a minute, and burning 128 bushes of coal per day, it dropped the level of water in the mine by nine feet a day, going as deep as thirty-five fathoms.[31]
Boulton’s business model for the firm was ingenious – rather than simply sell the engines, they would license them at a fee proportional the amount of coal saved. A mechanical meter devised by Watt and kept behind a locked door would count the engine’s strokes in order to determine the level of usage. The deal signed with Wheal Busy netted Boulton & Watt £700 per annum out of an estimated £2400 saved by the mine by using the new engine. Given this kind of economic dividend, it is not surprising that by 1780 Boulton & Watt had sold forty engines, half of them to mines in Cornwall. Just five years after the formation of their partnership, the Newcomen engine was all but eradicated there.[32]
But, for all the success of his new-and-improved design, Watt was far from done with the steam engine. Pumping water was all well and good, but vast territories of power still lay unconquered, and they would be unlocked by an engine that could do what a waterwheel did – generate circular motion.
[1] [Pope] Perhaps it was for the best, as a medieval Aristotilean likely would have considered the sentiment – which made a dangerously close analogy between Newton and God – blasphemous.
[2] Quoted in Friedrich Klemm, trans. Dorothea Waley Singer, A History of Western Technology (Cambridge, Mass.: MIT Press, 1964), 242-243.
[3] Ben Russell, James Watt: Making the World Anew (London: Reaktion, 2014), 48-52, 69-70. Smeaton quoted in Larry Stewart, “Science and the Eighteenth-Century Public,” in The Enlightenment World, P. Jones, et. al., ed. (Abingdon: Routledge, 2004), 240.
[4] Samuel Smiles, Lives of the Engineers, Volume II: Harbours – Lighthouses – Bridges. Smeaton and Rennie. (London: John Murray, 1874), 136.
[5] John Smeaton, “An Experimental Enquiry concerning the Natural Powers of Water and Wind to Turn Mills, and Other Machines, Depending on a Circular Motion,” Philosophical Transactions of the Royal Society 51 (1759), 105-106.
[6] The emergence of this new cultural synthesis between science and handicrafts is surveyed in Robert Friedel, A Culture of Improvement (Cambridge, MA: MIT Press 2010), 198-201.
[7] Richard L. Hills, Power from Steam: A History of the Stationary Steam Engine (Cambridge: Cambridge University Press, 1993), 37; Reports of the Late John Smeaton, F.R.S. vol. 2 (London: Longman, Hurst, etc., 1812), 348.
[8] Milton Kerker, “Science and the Steam Engine,” Technology and Culture 2, 4 (Autumn 1961), 381. This is not a direct quote of any historian, but rather Kerker’s summary of a certain point of view on science and the steam engine.
[9] We thus still use an ancient Greek conceptual frame when we talk about qualitative versus quantitative differences.
[10] Francis Bacon, Joseph Devey, ed., Novum Organum (New York: P.F. Collier & Son, 1902), 152-53.
[11] Thomas L. Hankins, Science and the Enlightenment (Cambridge: Cambridge University Press, 1985), 74-80.
[12] Hankins, 74-75.
[13] Russell, 81-84.
[14] Hankins, 75-76. The latent heat of steam was harder to measure, so Black’s figures for it not quite accurate.
[15] Hankins, 75. Johann Wilcke reached same conclusion independently in Sweden, and called this capacity “specific heat,” by analogy to the “specific gravity” that indicates a material’s density.
[16] Samuel Smiles, Lives of Boulton and Watt (London: William Clowes and Sons, 1865), 83-85; Irene Maver, “Eighteenth-century Glasgow,” Feb. 17, 2011, bbc.co.uk (https://www.bbc.co.uk/history/british/civil_war_revolution/scotland_glasgow_01.shtml)
[17] Smiles, 79-86.
[18] Smiles, 102-103; Russell, 41-45.
[19] Smiles, 108-113.
[20] Russell, 71-72.
[21] Russell, 73-77.
[22] The original engine on which the model was based had a 48-inch diameter cylinder. This would have had a wall somewhere between 1/3 of an inch and an inch thick. Scaling this down to a 2-inch diameter cylinder would have required a very thin and fragile wall (something like 1/50 of an inch). Instead, the maker used a wall about 5 times that thickness. Hills, 51-52.
[23] The degree to which Black or Watt should be credited for the improvement of the steam engine has been disputed, see Donald Fleming, “Latent Heat and the Invention of the Watt Engine,” in Otto Mayer, ed., Philosophers and Machines (New York: Science History Publications, 1976), 121-124. But for our purposes it hardly matters. Whether we credit Black for discovering latent heat or Watt for rediscovering it, both discoveries were due to the careful application of the new experimental philosophy.
[24] Russell, 84.
[25] Hills, 54-57.
[26] F. M. Scherer, “Invention and Innovation in the Watt-Boulton Steam-Engine Venture,” Technology and Culture
6, 2 (Spring, 1965), 168-170; Smiles, 153-157. As Scherer points out at 179, Watt’s overall design likely could have been made to work using Newcomen’s sealing strategy, had Watt been less of a perfectionist. It would have been more efficient than a Newcomen engine, but less efficient than the final Boulton & Watt engine.
[27] Scherer, 177-78. Watt also experimented with a variety of designs for the other components of his engine, as discussed at 179.
[28] Hills, 58-58; Soon after the boring machine’s precision was improved further by substituting a screw for the rack. E. A. Forward, “The Early History of the Cylinder Boring Machine,” Transactions of the Newcomen Society 5, 1 (1924), 24-38.
[29] Quoted in Scherer, 171.
[30] Russell, 109.
[31] Smiles, 235-36, 242-43; Hills, 59.
[32] Smiles, 234-49, 275.
