We last left the electric telegraph wandering through the decades in a kind of limbo. A fascinating demonstration piece, a promising curiosity, it had yet to prove itself as a practical instrument. By 1830, however, electricians had made several crucial new discoveries that made the electric telegraph as we know it possible. It began with a frog.
Animal Electricity
In November 1780, Luigi Galvani, physician and anatomist at the University of Bologna, was dissecting a frog with his ivory-handled scalpel. Nearby, an associate was playing with an electrical machine (what we would call an electrostatic generator). Suddenly, the frog’s leg convulsed. Galvani was perplexed – had he accidentally damaged the nerve? He did several more experiments, and discovered that the convulsions happened only when the electrical machine sparked while his finger touched the metal blade of the scalpel, which in turn was touching the frog.1
Natural philosophers had already known for some years that animals could produce electricity. The torpedo fish2, and its ability to benumb both fish and men, was known since ancient times, and described by Aristotle. The source of its power, though, was unknown for millennia. By the 18th century, though, some electricians began to suspect that its effect was electrical, having seen the similarly stupefying effect of the Leyden Jar. A series of experiments in the early 1770s finally proved beyond doubt the identity of the torpedo’s shock with electricity.3

Galvani’s experiments, however, had a more profound implication, that all animal life was in some fundamental sense electrical: “Electricity was… the great vital force, by which the decrees of the understanding, and the dictates of the will, were conveyed from the organs of the brain to the obedient members of the body.”4 This connection produced a general fascination in Europe with Galvani’s discoveries, a fascination whose most famous product was Mary Shelly’s Frankenstein.
More important for our purposes, however, was what Galvani found some six years later. Galvani’s nephew was hanging some frogs onto metal hooks hanging from the balcony outside his uncle’s lab, when he saw something strange. When the hooks touched the rail, the frogs convulsed continuously, exactly as if stimulated by an electric spark. The elder Galvani at first dismissed the anomaly, but then discovered that an even greater effect could be created by touching different metals to the frog at the same time, such as iron and brass or copper.5 This continuous flow of electricity, whose cause was still obscure, came to be known as galvanism.

Galvani believed he had found a kind of “animal electricity” – that the metal was connecting two separate reservoirs of electric fluid in the muscles and nerves, allowing them to flow, and thus generating nervous activity.6 His countryman, Alessandro Volta, was not convinced. Volta, a professor of physics at the University of Pavia near Milan, believed that the electrical flow came from the metal, not the animal, and set out to prove it.
So far, though, the only instruments found capable of detecting this ‘weak electricity’ from metal contact were frogs and human tongues. To prove his case Volta needed a non-animal detector. So he turned to the recently invented ‘doubler’ of William Nicholson, an English natural philosopher. By turning insulated brass discs past one another, the doubler could turn a small initial charge into an ever larger one. Using the doubler, Volta was able to build up the weak electricity from metal contact into something he could detect with an ordinary electrometer, disproving Galvani. But more curiously, he found that when rotated enough times the doubler by itself generated a detectable amount electricity, without any initial external charge.
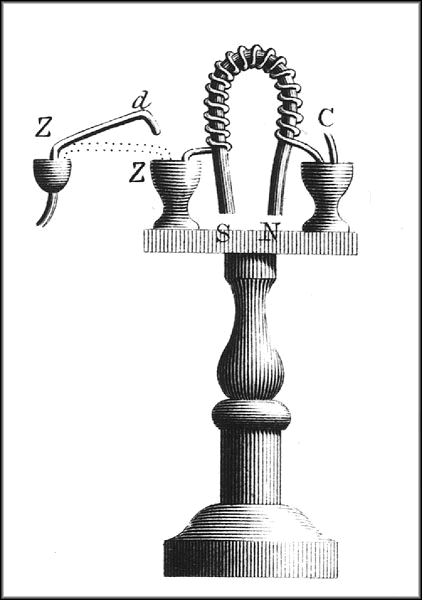
Soon Volta was experimenting with stacks of paired discs of various metals. He was inspired by another work of Nicholson’s, describing a dissection of the electric organ of the torpedo. Nicholson found within that organ a mass of columns, each concealing within a stack of tiny circular layers. Nicholson believed that a machine could be built to simulate the organ, perhaps with mica films. Volta agreed with the principle but differed on the materials. By 1799, he settled on a stack of paired zinc and copper discs, with pieces of brine-soaked cardboard or leather in between each pair. This device could by itself generate a significant and sustained electric flow. A second design, which he called “the crown of cups”, consisted of a series of cups containing acid or brine, connected by plates of two different metals soldered together.7

After Volta announced his discovery at the Royal Society in London in March of 1800, it spread like wildfire across Europe. Already by May Volta’s device had produced an important discovery – the decomposition of water by electricity – found by none other than Nicholson, working with the surgeon Anthony Carlisle. In English it came to be called a ‘battery’, a term already in common use to describe a combination of Leyden jars.
By this time Galvani the man was dead. In 1797, Napoleon reorganized northeastern Italy as the Cisalpine Republic, a French client state.8 Volta embraced the new order of things, received the Legion of Honor, and was made a count and a senator. Galvani, refusing to swear a loyalty oath to the republican government, was expelled from his post at the university and died in poverty the following year. The study of galvanism, however, continued to grow and thrive, with the aid of Volta’s battery.
Electro-magnetism
The battery made it possible, for the first time, to carefully study electricity in motion. The Leyden jar or other static electrical devices produced a single instantaneous jolt. But now a researcher could set up a battery, connect it to any given experimental apparatus, and study the effects of this dynamic electricity at leisure.
Researchers soon turned this new tool to the study of the relationship between electricity and magnetism. Since ancient times some had suspected a deep connection between these ‘occult’ attractive forces, but there were confounding differences as well (for example, electrified amber would attract any light object, the lodestone only iron).
New evidence came in the 17th century, when philosophers noted reports of lightning magnetizing iron objects (such as crosses atop church steeples) and reversing ship’s compasses. Ben Franklin showed that Leyden jars could likewise magnetize a needle or reverse a magnet – not surprising since by this time he and others had shown conclusively that lightning was electrical in nature. Still, the evidence was muddy. What exactly was the relationship between magnetism and electricity? Opinions at the end of the 18th century varied. Some though the apparent similarity coincidental, others were convinced that a single root cause explained both effects.9
After the discoveries of Galvani and Volta, philosophers began to explore possible interactions between galvanism and the magnet. The most important result came in 1820, when Hans Christian Ørsted, a professor of physics at the University of Copenhagen, noticed during a lecture that a galvanic current deflected a magnetized needle. After further experimentation, he published his findings that a current running from north to south above a needle would deflect its point to the west; placed below, to the east.10
When André-Marie Ampère read of this discovery in Paris, he began immediately to delve deeper. Ampère was a mathematics professor at the École Polytechnique – like the optical telegraph, a product of the French revolutionary state, intended in this case to provide military engineers who were steeped in cutting edge mathematics and physics.11 Where Oersted had noted the effect, Ampère sought the cause, applying his considerable mathematical and experimental skill to the task. He was able to cancel the effect of Earth’s magnetic field on the experiment by gluing together two needles with their poles in opposite directions. He found that this dual needle formed a precise right angle to the wire. Intrigued, he theorized that magnetism was caused by tiny circular currents of electricity, flowing at right angles to the magnetic north-south axis.
This model proved extremely fruitful over the following decade, as many others began experimenting with helical coils of wire in order to simulate these circular currents; first of all his colleague François Arago, who showed that such a coil could permanently magnetize a needle.
A few years later, Scotsman William Sturgeon did Arago one better. He was looking for ways to make powerful and striking electro-magnetic effects with a small battery, in order to demonstrate electromagnetic principles more easily to students and the general public. As it was, copious amounts of galvanic power, from a large, expensive, and difficult to manage battery, were required to make small magnetic effects, barely visible to all but the closest observers.
Sturgeon found that by wrapping a wire around a lacquered bar of iron, and attaching it to a small battery, he could make a powerful electro-magnet, which retained its magnetism as long as electrical current flowed through the wire. The magnet became even stronger when he bent the bar into a horseshoe shape, and was capable of lifting as much as nine pounds.12
Joseph Henry, a teacher at the Albany Academy in Albany, New York, kept up with European work in electricity and magnetism from journals such as the Annals of Philosophy, where Sturgeon published a description of his electro-magnet.
Henry was also trying to make better equipment for pedagogical demonstrations, but he believed he could do better than Sturgeon by taking Ampère’s model of circular currents to the limit. To do so, Henry hybridized Sturgeon’s electro-magnet with German Julian Schweigger’s ‘multiplier’, which used tightly-wound silk-wrapped wire to greatly amplify the effect of Oersted’s experiment.

Using wire insulated with silk in this way allowed Henry to pack his coil of wire tightly around the iron core without any risk of a short, better approximating Ampère’s circular currents than Sturgeon’s loose helix. With this improvement, he found he could lift hundreds, and later thousands, of pounds, all with an ordinary battery.13
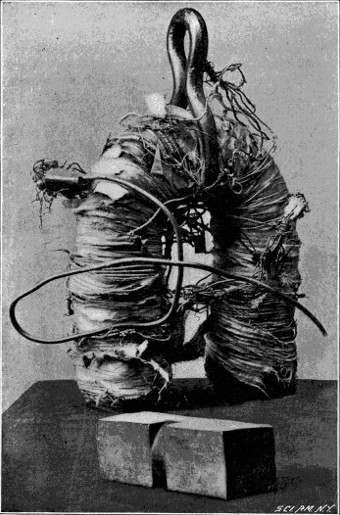
This ability of electricity to generate magnetism, and thus draw metal to itself – long suspected, at last proven by Ørsted, and developed to perfection by those who followed in his footsteps – would prove of tremendous value to the builders of electric telegraphs.
A Theory of Circuits
At this point it was not hard to imagine a new kind of telegraph, using the voltaic battery as its source and the electromagnetic effect for its detector. Ampère himself pointed this out as early as 1820.
In 1824, Peter Barlow, a renowned mathematics professor at the Royal Military Academy near London, decided to investigate whether a telegraph such as that described by Ampère could in fact work over long distances. To his disappointment, he found that the strength of the electromagnetic effect diminished greatly with as little as 200 feet of wire, convincing him that an electromagnetic telegraph could never be practical.14 Given Barlow’s reputation, this was enough to discourage further telegraphic experimentation in England for over a decade.
Why had some experiments with electric telegraphy failed as utterly as Barlow’s, while others (like Francis Ronalds’) succeeded through miles of wire? An intellectual framework was needed, a model for understanding the behavior of electricity in motion, in order to determine what kind of apparatus would work and what would not.
Here Joseph Henry once again enters our story. In his experiments with the electromagnet in the late 1820s, Henry found that there was a point of diminishing returns – at some point the loss of strength from the length of the wire defeated any benefit from adding additional turns around the core. This, by itself, simply confirmed Barlow’s conclusion.

But his experiments went further. First, he found that by splitting a long wire into multiple pieces, each wrapped around the iron core and then attached separately to the battery – what we would call a parallel circuit – he could overcome this limitation and create extremely powerful electromagnets. Then he went back to his single wire magnet and tried attaching it to a different kind of battery. Instead of a single simple cell with two large metal plates bathed in acid, he used a wooden trough lined with 25 small cells, each linked to the next. To his surprise, it lifted slightly more weight through 1/5 mile of wire than when attached directly to the battery!
Henry intuited that there were two different modalities of electricity at work here, quantity and intensity. A quantity battery with a single large cell paired with a quantity magnet, wrapped in multiple wires, and this pairing excelled at generating large amounts of mechanical force. On the other hand, an intensity battery with many cells paired with an intensity magnet with a single coil of wire, and this pairing excelled at transmitting the electromagnetic effect through long lengths of wire. Henry was well aware of the practical implications of this work, noting in his paper that it was “directly applicable to Mr. Barlow’s project of forming an electro-magnetic telegraph.”15
It would not have been polite of Henry to say simply that he had proved Mr. Barlow wrong, but he showed as much by stringing up a demonstration circuit at the Albany Academy that could ring a bell through a circuit a mile and a half long, using an intensity battery. His model also explained why Ronalds’ telegraph had worked at long distances – the frictional electrostatic generator that Ronalds used was an inherently high-intensity device.
Though he would not learn of it until a tour abroad in 1837, Henry’s intuitive model had in fact already been rigorously mathematized, by Georg Ohm, a Bavarian by birth who was at the time a physics and mathematics teacher in Prussia.

Combining thorough experimentation with mathematical imagination, Ohm carefully studied the strength of various circuits, as measured by Schweigger’s multipler (by then commonly called a galvanometer). He concluded that the strength of the current (Ströme – Henry’s quantity) of a circuit depended simply on its tension (Spannung – Henry’s intensity) divided by its ‘reduced’ length (that is, normalized against a standard wire).16
Ohm’s model of the circuit was far more sophisticated than Henry’s, and was published four years earlier, in 1827. In that sense his publication should have rendered Henry’s moot. But Ohm’s work was not well-received in Prussia, and so did not become widely known in the scientific world until the 1840s. Moreover, unlike Ohm, Henry explicitly presented his findings as a solution to the electric telegraph’s distance problem. It was by no means obvious that a solution also lurked in Ohm’s mathematics. And so Henry’s analysis of quantity and intensity would have a decisive effect on telegraphic developments.17
The Last Mile
The battery and the new form of ‘galvanic’ electricity that it engendered transformed work on telegraphy. To begin with, it was a new source of electricity, much more consistent and tame than that from electrostatic machines, and could be put to useful work. But indirectly it also made electricity vastly easier to experiment with, enabling, in turn, the discovery of electromagnetism and a deeper understanding of circuits.
Early telegraphic detectors depended on the chemical effects of electricity or its relatively weak attractive powers – it could lift a piece of paper, but little more. The electromagnet transformed electrical energy into a brute mechanical force, opening up far greater flexibility in the design of telegraphic equipment. Henry and Ohm’s theories of currents then provided instruction on which arrangements of equipment would prove fruitful, and which not.
The core ideas were now available for the construction of the electromagnetic telegraph as we know it. It remained for those with the necessary will and resources to assemble them, and transform them into a useful system. [Next Part]
Further Reading
John J. Fahie, A History of Electric Telegraphy to the Year 1837 (1974 [1884])
Bruce J. Hunt, Pursuing Power and Light : Technology and Physics from James Watt to Albert Einstein (2010)
Guilliano Pancaldi, Volta: Science and Culture in the Age of Enlightenment (2003)
- John J. Fahie, A History of Electric Telegraphy to the Year 1837 (1974 [1884]), 180-181. ↩
- The name of the torpedo comes from the latin word for numbness or paralysis, which also gives us torpor. Its name, in turn, was given to an underwater weapon. With perhaps some resentment on the part of the fish, who after all has to eat, but doesn’t go about sinking ships full of people willy-nilly. ↩
- Fahie, 169-73. ↩
- Fahie, 185. ↩
- Fahie, 181-83. ↩
- Fahie, 182-83. ↩
- Guilliano Pancaldi, Volta: Science and Culture in the Age of Enlightenment (2003), 180-89. ↩
- The name was a classical allusion: The Romans dubbed the lands of the Gauls in Italy Cisalpine Gaul, as opposed to Transalpine Gaul across the Alps, which roughly corresponds to modern France. ↩
- Fahie, 250-6. ↩
- Fahie, 273-4. ↩
- It would later serve as a model for West Point in the U.S. (http://www.uh.edu/engines/asmedall.htm). ↩
- William Sturgeon, “Account of an improved electromagnetic apparatus”, in Annals of Philosophy, vol. 12 (1826), 357-361. ↩
- Albert E. Moyer, Joseph Henry: The Rise of an American Scientist (1997), 61-67. ↩
- Peter Barlow, “On the Laws of Electro-Magnetic Action as depending on the Length and Dimensions of the conducting Wire and on the question Whether Electrical Phenomena are due to the transmission of a single or of a compound fluid?”, Edinburgh Philosophical Journal, vol. 12 (1825), 105–113. ↩
- “On the application of the principle of the galvanic multiplier to electro-magnetic apparatus, &c,” Sillimans’ American Journal of Science, January 1831; in The Scientific Writings of Joseph Henry, vol. 1 (1886), 37-49. ↩
- Georg Simon Ohm, Die Galvanische Kette: Mathematisch Bearbeit (1827), 178. ↩
- E.A. Marland, Early Electrical Communication (1964), 50. ↩

Hi thanks foor posting this
LikeLike